Rubella part 3
The Rubella pandemic of 1962-1965, with about 12.5 million cases, resulting in 2,000 cases of encephalitis, 2,100 neonatal deaths, 20,000 infants born with Congenital Rubella Syndrome, and 11,250 therapeutic or spontaneous abortions, inspired the development of the Rubella vaccine, principally targeting the reduction of Congenital Rubella Syndrome.
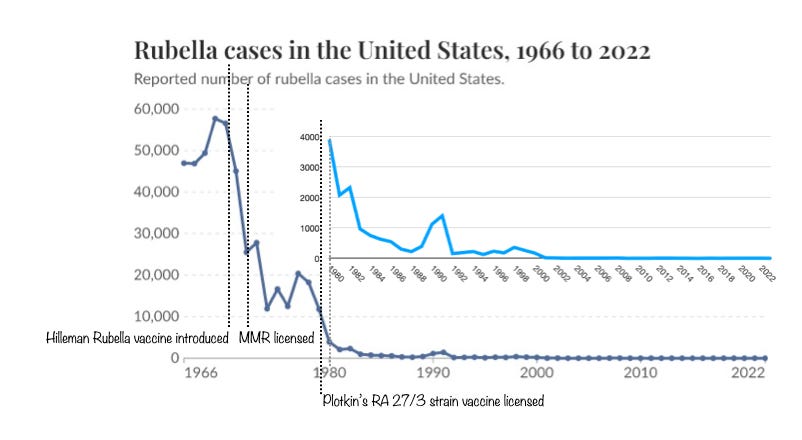
The Rubella virus was isolated in 1962, and the first Rubella vaccine was licensed in the U.S. in 1969, developed by Maurice Hilleman, who also developed vaccines for measles and mumps.
Stanley Plotkin developed the RA 27/3 rubella vaccine strain, attenuated by passage in WI-38 human diploid fibroblasts, used in all contemporary rubella vaccines, at the Wistar Institute in Philadelphia in the wake of the 1964-1965 epidemic. Vaccine based on the RA 27/3 strain, determined to induce higher antibody titers and produce an immune response more closely paralleling natural infection than the Hilleman vaccine, was licensed in 1979.
Rubella is included in the MMR, recommended at 12-15 months & 5 years of age. A single dose of the RA 27/3 vaccine in current use is ~97% effective in preventing rubella infection, and provides sterilizing immunity, impacting transmission.
Following the introduction of vaccination, Rubella cases plummeted from 57,686 in 1969 to 225 in 1988. More significantly, Congenital Rubella Syndrome incidence fell from 68 reported cases in 1970 to 19 in 1974.
In 1979, following a spike in Congenital Rubella Syndrome cases, the CDC recommended vaccination of young adults (over 18) without evidence of Rubella immunity (documented history of natural disease or negative serology), including postpartum vaccination for women with negative serology in prenatal testing. As the rubella vaccine is an attenuated live virus vaccine, its use in pregnancy is not recommended on theoretical grounds, tho inadvertent vaccination in unidentified pregnancies has not been seen to result in adverse outcomes.
The rationale for vaccination of children is not so much to prevent childhood disease, which is relatively inconsequential, but to generate population immunity to reduce community transmission in order to avoid exposure of women in pregnancy.
The estimated R0 for rubella is 5-7, suggesting that the “herd” (community) immunity threshold to prevent epidemic transmission is between 80-86%, more easily accomplished than for measles.
Rubella immunity from natural exposure or vaccination is typically lifelong, with immunity based on long-lived memory B & T cells, but circulating antibody levels from long-lived plasma cells can wane over 15–17 years, at 2.6% to 8.2% annually, leading to susceptibility in young adults, including in women of childbearing age and their adult contacts, more concerning re the transfer of passive immunity to the fetus in pregnancy. This was the case as well for naturally acquired immunity in the pre-vaccine era, although periodic exposure to Rubella circulating in the community likely “boosted” antibody production. Rubella immunoglobulin titres are routinely obtained in early pregnancy, with vaccination recommended following delivery to protect future pregnancies, and vaccination is recommended for young adults of childbearing age with negative serology outside of pregnancy. The most effective strategy to protect non-immune women in pregnancy is to assure vaccination of the community at large, to prevent community transmission.
Genomic sequencing of US and international rubella viral isolates has been ongoing since 1996, with historical isolates analyzed as far back as 1957. 2 clades and 13 genotypes have been identified, all belonging to a single stable serotype. As with measles, these cannot be considered strains or variants, as they do not present differences in virulence, antigenicity, or transmissibility; genotyping is of interest principally in tracing chains of transmission. The primary antigenic target for neutralizing antibodies is the E1 glycoprotein, which mediates membrane fusion and is highly conserved across all genotypes due to functional constraints.
Only 2 cases of Congenital Rubella Syndrome have been reported in the U.S. over the past 20 years, both in 2008.
The RA 27/3 rubella vaccine strain was attenuated by passage in WI-38 human diploid fibroblasts obtained from a single electively aborted 3-month-gestation fetus in Sweden in 1962. This is sometimes misrepresented to suggest erroneously that the vaccine “contains fetal cells.” It does not. Fetal cells were used in the attenuation of the virus, but are not present in the vaccine. For those who respect the authority of the Roman Church, in 2005, the Vatican Pontifical Academy for Life issued a document Moral Reflections on Vaccines Produced from Cells Derived from Aborted Human Foetuses, stating “The Pontifical Academy for Life rejects the claim that Catholics have a moral duty to refuse the rubella vaccine on the grounds of conscience and Catholic teaching. It encourages Catholic parents to vaccinate their children against rubella and other serious diseases despite the unfortunate origin of the cell lines used in the manufacture of the vaccines.”