Measles immune amnesia
Let’s first examine the elements of acquired immunity
Immune globulins - immune globulins produced in reaction to infection typically have a half-life of ~21 days; IgM in the acute phase of infection, IgA on the mucosa, IgG in convalescence as the most important effector in near-term immune memory, in some cases persisting in circulation for years.
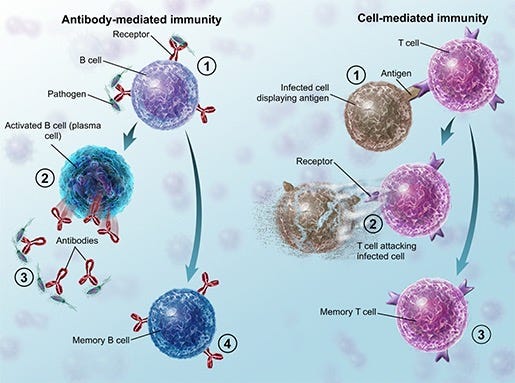
Long-term acquired (adaptive) immunity is dependent on long-lived effector B & T cells, & on memory B- & T-cells.
Plasma cells (effector B cells) - antigen-specific activated B lymphocytes - are the cells that generate immune globulins. These are of two classes:
short-lived plasma cells reside in secondary lymphoid tissues, producing large quantities of antibodies, typically persisting for only 3–5 days after activation before undergoing apoptosis.
long-lived plasma cells reside in the bone marrow and continue to secrete antibodies for decades or even for the lifetime of an individual independent of antigen restimulation, providing long-term protection.
Memory B cells - derived from antigen-specific effector B cells, can survive for decades in regional lymphoid tissues and undergo clonal proliferation on antigen restimulation, generating fresh clones of antigen-specific effector B cells much more rapidly and robustly than the primary adaptive immune response. Memory B cells can undergo class-switching of immunoglobulin type, generating an assortment of IgM, IgG, and IgA antibodies.
T effector cells, including CD8+ Cytotoxic T Cells (”killer T cells”) and CD4+ Helper T Cells are typically short-lived, undergoing apoptosis after the pathogen is cleared.
Antigen-specific Central Memory T cells persist for years in lymphoid organs, and Effector Memory T cells persist in peripheral tissues for years, undergoing clonal proliferation, generating clones of antigen-specific effector T cells on antigen restimulation, again, far more rapidly than the primary adaptive immune response.
Maternally-acquired Effector B- & T- cells and memory B- & T-cells, specific to pathogens experienced historically by the mother, transferred transplacentally & via colostrum & breastmilk, may persist for years in the lymphoid tissues as materno-fetal microchimerisms.
Long-lived plasma cells and Memory B- & T cells, including those involving maternal microchimerisms, essentially constitute a library of information regarding historical infections, with long-lived plasma cells providing immediate protection and memory B- & T-cells capable of being brought into play within 2-4 days of exposure to a pathogen, while the primary adaptive immune response requires ~1–2 weeks to mount a response to infection.
Wild type measles virus infects lymphocytes bearing the CD150 (SLAMF1) receptor, including B effector cells, effector T cells, and B & T memory cells, inducing apoptosis (death) of these cell populations, resulting in both short-term immunosuppression and long-term immune amnesia, effectively erasing between 11-73% of a child’s protective immune memory, requiring a primary immune response from re-exposure to pathogens to regain protective immunity, resulting in increased susceptibility to all-cause infectious disease. Measles vaccination, by preventing this, protects individual and population polymicrobial immunity. (The attenuated vaccine strain virus uses the CD46 receptor for cellular entry rather than CD150/SLAM, does not infect lymphocytes, and does not induce immune amnesia). Measles vaccination has been observed to reduce all-cause mortality more than explained by prevention of measles infection per se, by reducing the impact of measles-induced immune amnesia to other pathogens.